Acetone Polar or Nonpolar Solvent
Nonpolar solvents include things like toluene gasoline and oils. Apolar or nonpolar solvents have low dielectric constants which refers to the electric charge between the molecules being evenly distributed.
Polar Vs Nonpolar Solvents Identifications And Examples Psiberg
The polarity of lipids can be different.
. Water-free synthesis of Ti 3 C 2 T x MXene in ammonium dihydrogen fluoride NH 4 HF 2 dissolved in polar organic solvents eg propylene. Polar lipids such as glycolipids or phospholipids are more soluble in polar organic solvents such as alcohols. It dissolves a wide range of both polar and nonpolar soils and is often used to dissolve and remove light oils fingerprints cutting fluids flux residues carbon deposits and mold release.
Typically the solvent is what everything else is dissolved into. Because the solvent transports the chemical along the plate the solvent utilised has a significant impact on the chemicals retention factor value. Acetic acid undergoes nearly all carboxylic acid reactions.
Polar solvents are often found to have a high dielectric constant although other solvent scales are also used to classify solvent polarity. In contrast in eg. The structure of the fatty acids determines whether or not the fat is.
A solvent is typically thought of as liquid and that is the most common form. It is common to try three to six solvent systems for the first round of. Solvent Impacts Retention Factors.
The analytes the solvent front and the point where the mixture AB was administered are all measured relative to each other. Let this beaker sit beside the water beaker. The nonpolar layer is referred to as the organic phase and will dissolve any nonpolar compounds.
These are solvents having a dielectric constant of more than 15. A heterocyclic organic compound. A non-polar solvent will dissolve non-polar molecules.
This is particularly true for nonpolar solvents. They found that the dissolution rate decreases with increasing solvent size indicating that dissolution rate is limited by the rate of which solvent molecules penetrate. Many nonpolar solvents fall into the category of volatile organic compounds VOCs and are quite dangerous.
Polymers formed by a straightforward linking together of monomer units with no loss or gain of material are called addition polymers or chain-growth polymersA listing of some important addition polymers and their monomer precursors is presented in the following table. The polarity differentiation is done based on the dielectric constant of the solvents. Isopropyl alcohol CAS 67-63-0 is also referred to as IPA isopropanol 2-propanol and even rubbing alcohol more on that later.
Also any potential hydrogen-bond acceptor will tend to shift the water signal down-field. Sodium chloride has a NaCl molecule which breaks into Na and Cl- ions when dissolved in water. It can also be a solid or gas.
A powerful psychoactive drug. Many polymeric materials having chain-like structures similar to polyethylene are known. They can dissolve salts and other ionizable solutes.
It should be noted that the latter is quite temperature-dependent videinfra. The conductivity of a solution depends on the solvation of its ions. For binary mixtures of acetoneisopropanol a transition from swelling to dissolution occurred near acetone volume fractions of 04505.
Nonpolar solvents cannot solvate ions and ions will. Add a nonpolar solvent to a new beaker. Polar solvents can be used to dissolve inorganic or ionic compounds such as salts.
Owing to its ability to dissolve both polar and nonpolar compounds acetone is a strong solvent while other solvents can only dissolve one or the other. Ethers are a common example of organic phases in extractions. 08 then add more nonpolar solvent or switch to an even less polar combination such as pentaneether.
Waxes steroids phospholipids and fats are the most common types of lipid groups. Lipids as a class of compounds are insoluble in water but are soluble in other organic solventsExamples of such solvents include acetone and ether. And nonpolar gases such as ammonia ethanol and acetone at room temperature mostly because of their metallic core channels and surface functional groups that result in strong adsorption energies.
Acetic acid is a polar protic solvent with a dielectric constant of 62 in its liquid form. Acetic acid undergoes decomposition when heated above 440C to yield either methane and carbon dioxide or water and ethanone given by the equations. Fats have glycerol in addition to three fatty acids.
The polar layer is referred to as the aqueous phase and it will dissolve any polar compounds. Additionally solutes will be more soluble if the molecules in the solute are smaller than the ones in the solvent. The most common three types of solvents in organic chemistry are apolar polar aprotic and polar protic.
Water is a common example of an aqueous phase in extractions. Add 100 mL of a chosen nonpolar solvent to another beaker. That is solutes typically will dissolve best in solvents that have the most molecular similarities.
Why is acetone a better solvent than water. DMSO the water is already strongly. Secondly since it is miscible acetone is a strong solvent ensuring it has the potential to blend in all amounts with water.
The efficiency of the extraction depends on the polarity of the lipids and polarity of the solvent Ramluckan et al 2014. Polar solutes will dissolve better in polar solvents and nonpolar solutes will dissolve better in nonpolar solvents. Water as a solvent dissolves sodium chloride by breaking into ions.
Used in alcoholic beverages in thermometers as a solvent and as a fuel Fehlings reagent. An important polar aprotic solvent that dissolves both polar and nonpolar compounds Dioxane. There can be both polar and nonpolar solvents.
Generally a polar solvent will dissolve polar molecules. Factors Affecting R f Values 1. The ideal solvent for lipid extraction must be selected.
Shifts of the solvent residual peak2 and the water peak. Classified as an ether Ethanol.

Is C3h6o Acetone Polar Or Non Polar Youtube
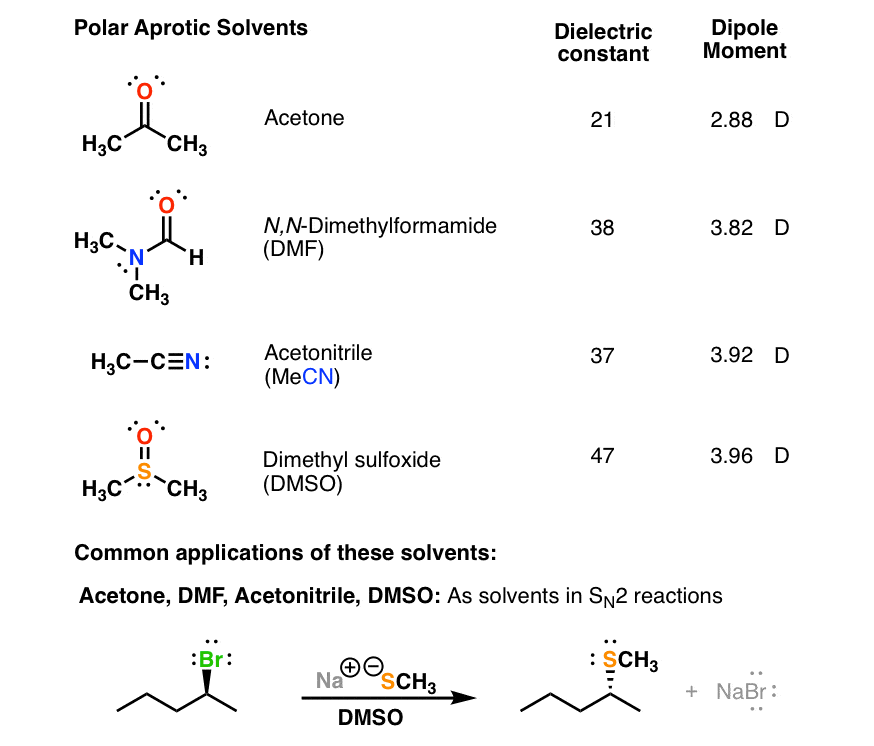
Polar Protic Polar Aprotic Nonpolar All About Solvents

Polar Protic Polar Aprotic Nonpolar All About Solvents
What Makes Acetone A Really Good Solvent What Allows It To Dissolve Both Polar And Non Polar Molecules Quora
0 Response to "Acetone Polar or Nonpolar Solvent"
Post a Comment